Researchers from the University of Maine and Penn State have discovered that molecules can experience non-reciprocal interactions without the need for external forces.
For instance, predators are attracted to prey, while prey tends to flee from predators. These non-reciprocal interactions play a crucial role in the complex behavior observed in living organisms. In the case of microscopic systems like bacteria, non-reciprocal interactions have been explained by hydrodynamic or other external forces. It was previously believed that similar forces could explain interactions between individual molecules.
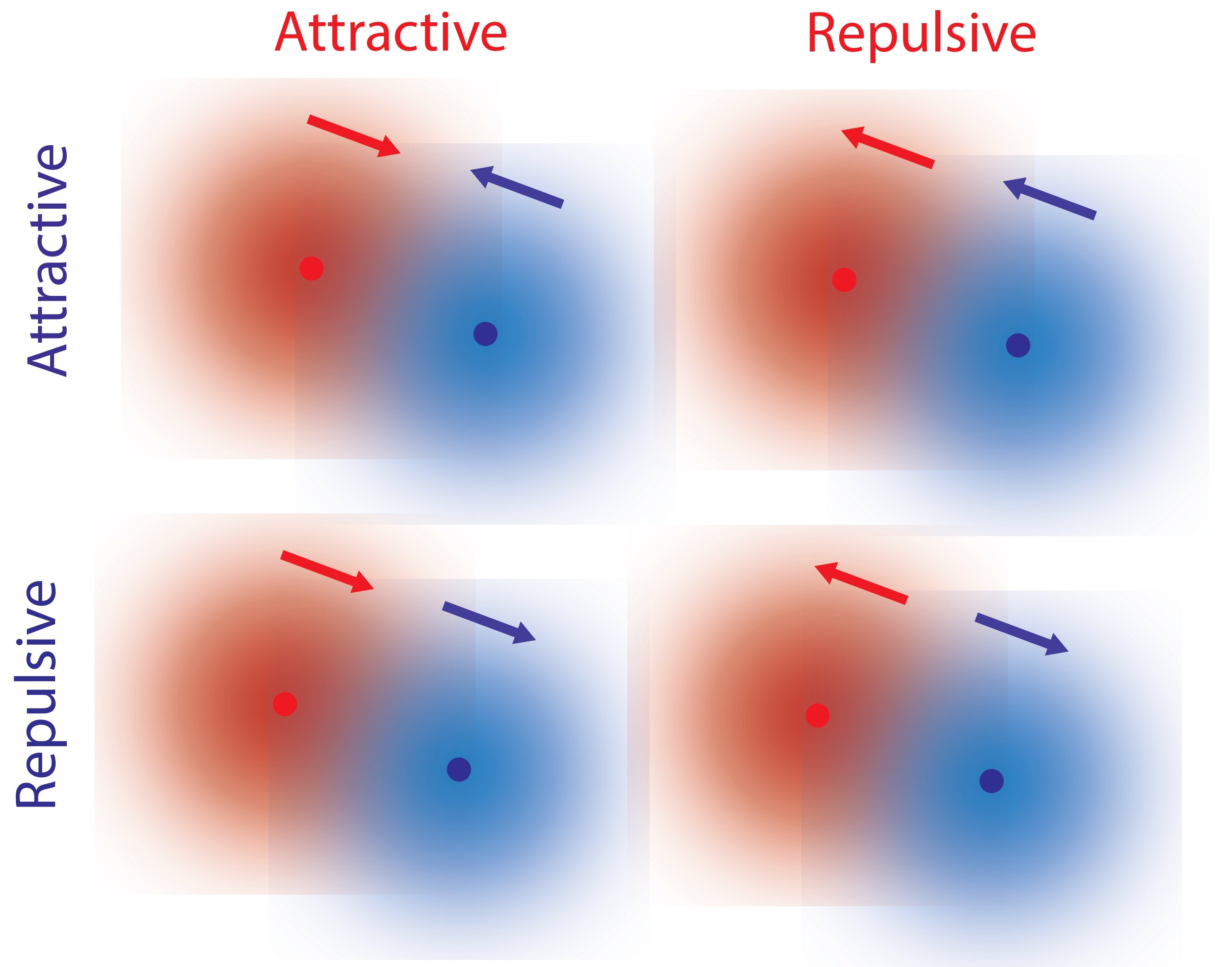
In a study published in Chem, theoretical physicist R. Dean Astumian from UMaine and collaborators Ayusman Sen and Niladri Sekhar Mandal from Penn State propose a different mechanism by which single molecules can interact non-reciprocally without hydrodynamic effects.
This mechanism involves the local gradients of reactants and products resulting from reactions facilitated by chemical catalysts, such as enzymes in biological systems. Depending on the properties of the catalyst, it is possible for one molecule to repel while attracting another molecule.
The researchers had a “Eureka moment” during their discussion when they realized that the kinetic asymmetry, a property of every catalyst, controls the direction of response to a concentration gradient. Since kinetic asymmetry is an inherent property of enzymes, it can undergo evolution and adaptation.
2023-12-31 19:00:04
Source from phys.org