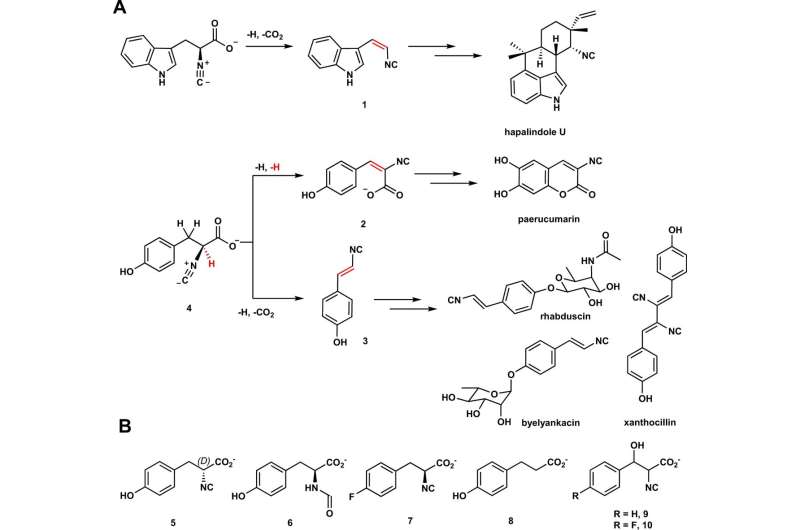
A) Hapalindole U, paerucumarin, rhabduscin, and byelyankacin are biosynthesized by way of 1, 2, or 3 as key intermediates. Iron- and 2-oxoglutarate-dependent enzymes catalyze decarboxylation-assisted desaturation and formal dehydrogenation to put in vinyl isonitrile (1 and three) and isocyanoacrylate (2) teams, respectively. The place of desaturation is highlighted in crimson. B) Substrate analogs (5–8) and mechanistic probes (9–10) used on this examine. Credit: Wantae Kim et al, Nature Communications (2022). DOI: 10.1038/s41467-022-32870-4
Researchers from North Carolina State University and the University of Texas at Austin have outlined the construction of a substrate-bound iron 2-oxoglutarate (Fe/2OG) enzyme to discover whether or not these enzymes might be used to create a wide selection of molecules. They probed the enzyme’s lively web site to find out its potential to bind with totally different substrates. Additionally, slightly than oxygen-addition, they noticed that Fe/2OG enzymes possible make the most of cations—extremely reactive species—to drive desaturation throughout catalysis. The work, revealed in Nature Communications, may result in using Fe/2OG enzymes in making a wide selection of helpful molecules.
Enzymes within the Fe/2OG household are naturally occurring—they’re present in every thing from micro organism to crops and animals. As such, these enzymes have the potential for use as a greener, extra environment friendly platform for creating molecules equivalent to vinyl isonitriles, which have antibiotic properties. However, the pathways by which Fe/2OG enzymes create these molecules are poorly understood.
“The endgame is to grasp how the enzymes on this household create specific molecules, in order that we will doubtlessly piggyback on a pure course of that present chemistry can not replicate,” says Wei-chen Chang, affiliate professor of chemistry at North Carolina State University and co-corresponding creator of a paper describing the work. “So we checked out a few totally different enzymes throughout the Fe/2OG household to see how they carried out totally different transformations utilizing the identical substrate, or molecule they bind to.”
By specializing in how the enzyme binds to a specific substrate, the researchers can decide which different substrates might be utilized by the enzyme, a extra environment friendly strategy to decide potential reactions and merchandise than experimentation.
The analysis group targeted on two Fe/2OG enzymes—PvcB and PlsnB—and in contrast their constructions and the merchandise they made. They recognized binding websites on each enzymes, however when it got here to exploring how the enzyme carried out its transformations, they made a shocking discover.
“Normally, the best way Fe/2OG enzymes catalyze or create a brand new product occurs like this: the enzyme binds to the substrate, a single oxygen atom from molecular oxygen (O2) is launched into the substrate, and oxygen-addition drives the response,” Chang says. “That course of known as hydroxylation.
“But for these enzymes, the transformation or response is not pushed by hydroxylation, however by a reactive cation that triggers the next desaturations, the place new bonds are launched.”
The two Fe/2OG enzymes studied (PIsnB and PvcB) utilized essentially distinct desaturation to create totally different merchandise from the identical substrate.
“Now we all know how these enzymes catalyze transformations and have discovered the binding websites, we now have a basis for figuring out what they will do by way of reactions,” Chang says. “We may suggest and predict the perfect substrates to make use of to get focused merchandise.”
Optimal concentrations of enzymes and their substrates
More data:
Wantae Kim et al, Elucidation of divergent desaturation pathways within the formation of vinyl isonitrile and isocyanoacrylate, Nature Communications (2022). DOI: 10.1038/s41467-022-32870-4
Provided by
North Carolina State University
Citation:
Researchers discover enzymes that use a cation, not oxygen-addition, to drive reactions (2022, September 12)
retrieved 12 September 2022
from https://phys.org/information/2022-09-explore-enzymes-cation-oxygen-addition-reactions.html
This doc is topic to copyright. Apart from any honest dealing for the aim of personal examine or analysis, no
half could also be reproduced with out the written permission. The content material is offered for data functions solely.